공지/교육
법령
[Chemlinked] EU SCCS Consults on Opinions About Butylparaben and Methylparaben
첨부파일
등록일 2023-06-19
조회수 5611
On June 8, 2023, EU Scientific Committee on Consumer Safety (SCCS) published preliminary opinions respectively on the use of Butylparaben (CAS No. 94-26-8) and Methylparaben (CAS No. 99-76-3) in cosmetics. The opinions are open for comments until August 21, 2023.1-2
Endocrine disruptors (EDs) are chemical substances that can alter the functioning of the endocrine system, and consequently produce adverse effects upon human health. In terms of the management of EDs, the Regulation (EC) No 1223/2009 (Cosmetics Regulation) does not contain specific provisions. Instead, substances identified as EDs, including Butylparaben and Methylparaben, are currently subject to the general safety assessment of SCCS. To ensure their safe use in cosmetics, the European Commission requested SCCS to conduct safety assessments on Butylparaben and Methylparaben.
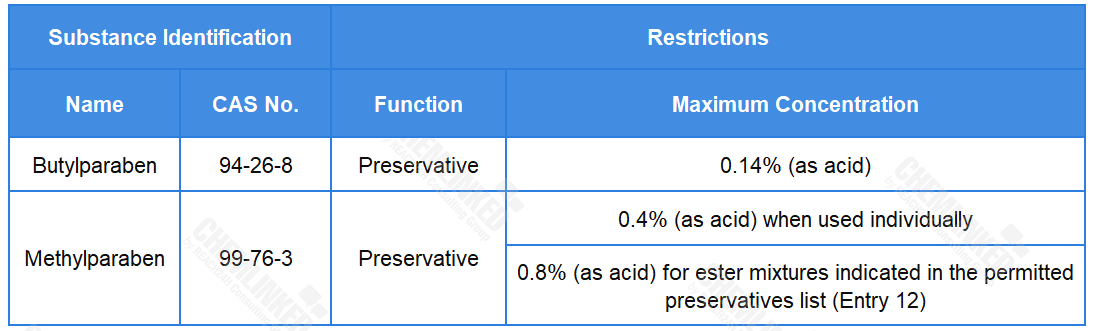
Based on the assessments of all available information, including the potential endocrine effects, SCCS considers the use of Butylparaben and Methylparaben in cosmetics is safe as shown below:
Further Reading
- New EU Regulation on General Product Safety to Take Effect in 2024
- EU Adopts the 12th Revision of The SCCS Notes of Guidance for the Testing of Cosmetic Ingredients and Their Safety Evaluation
- EU to Amend the Use Requirements for 13 Nanomaterials in Cosmetics
Reference Links
[1] SCCS—Preliminary Opinion Open for Comments on Butylparaben (Deadline: 21 August 2023)
[2] SCCS—Preliminary Opinion Open for Comments on Methylparaben (Deadline: 21 August 2023)
출처 : Chemlinked








